England: A ‘game-changing’ HIV-prevention treatment has been approved for use in England and Wales. Cabotegravir (CAB-LA), an injection given every two months.
It is designed to reduce the risk of infection in HIV-negative adults and young people, offering a significant alternative to the daily PrEP (pre-exposure prophylaxis) pills. Some individuals cannot take PrEP due to medical or other reasons, making CAB-LA a crucial option.
The National Institute for Health and Care Excellence (Nice) issued draft guidance recommending the use of CAB-LA, marking it as the first injectable PrEP option and the first PrEP medicine to receive Nice approval.
NICE approves first HIV prevention injection for those who can't take daily tablets.
Cabotegravir offers a new PrEP option for high-risk individuals, used alongside safer sex practices: https://t.co/zUegv7VtcJ pic.twitter.com/a5XFqdEWWn
— NICE (@NICEComms) October 17, 2025
The treatment is expected to be available roughly three months after Nice publishes its final guidance later this year. Health Secretary Wes Streeting described the approval as ‘game-changing’ and emphasized that the CAB-LA injection represents hope for vulnerable people who cannot use other HIV prevention methods.
Streeting added that, “We’re making real progress on HIV, with PrEP use up by 8 percent this year, and our ambition goes even further. England will be the first country to end HIV transmissions by 2030, and this breakthrough treatment is another powerful tool in our arsenal to reach that crucial goal.”
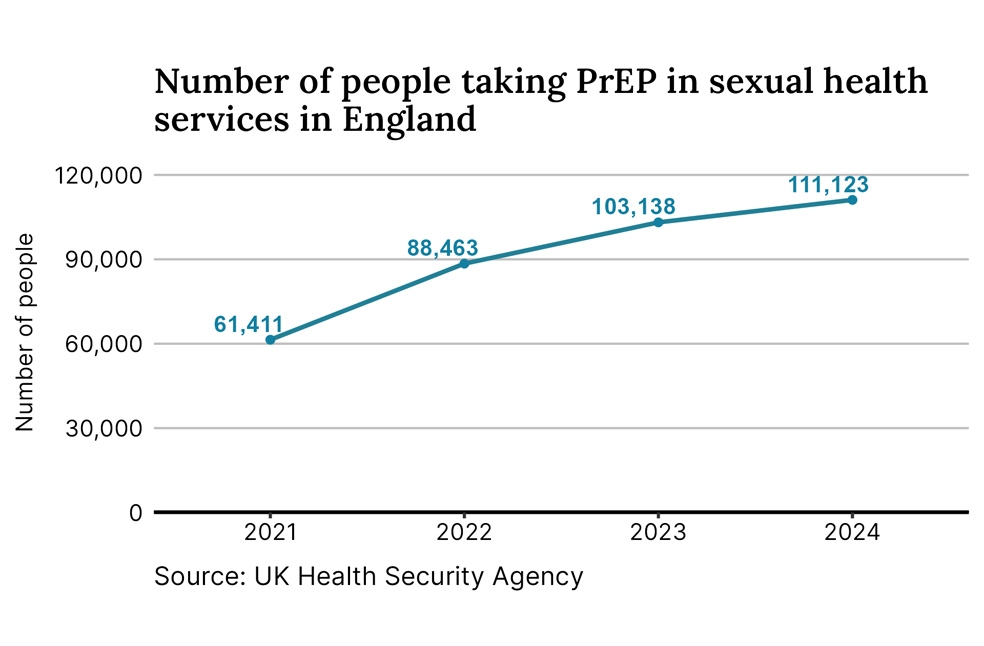
According to the UK Health Security Agency (UKHSA), more than 111,000 people accessed PrEP in sexual health clinics in England last year, a 7 percent increase compared to the previous year.
Helen Knight, director of medicines evaluation at Nice, noted that HIV remains a serious public health challenge, but CAB-LA provides a powerful tool to prevent new infections.
Knight explained that around 1,000 people in England cannot take daily oral PrEP due to medical contraindications or other barriers, and this injectable treatment offers an effective alternative. Approximately the same number of people are expected to benefit from the new treatment in England each year.
The approval of CAB-LA shot reinforces ongoing efforts in England and Wales to expand HIV prevention options, provide greater accessibility for vulnerable populations, and advance toward the goal of ending HIV transmissions by 2030.