London: Several LED face mask adverts have been banned by the Advertising Standards Authority (ASA) for making unapproved claims that the devices can treat medical skin conditions such as acne and rosacea.
The surge in popularity of at-home beauty devices, fuelled by social media influencers and online reviews, has prompted increasing scrutiny from regulators. While LED therapy is believed to stimulate skin cells and improve appearance, only devices registered with the Medicines and Healthcare products Regulatory Agency (MHRA) are legally allowed to make medical claims.
Unproven claims and regulatory action
The ASA used artificial intelligence to detect adverts that might breach advertising rules. The watchdog found that several companies promoted LED masks with claims of treating acne and rosacea, despite not being registered as medical devices.
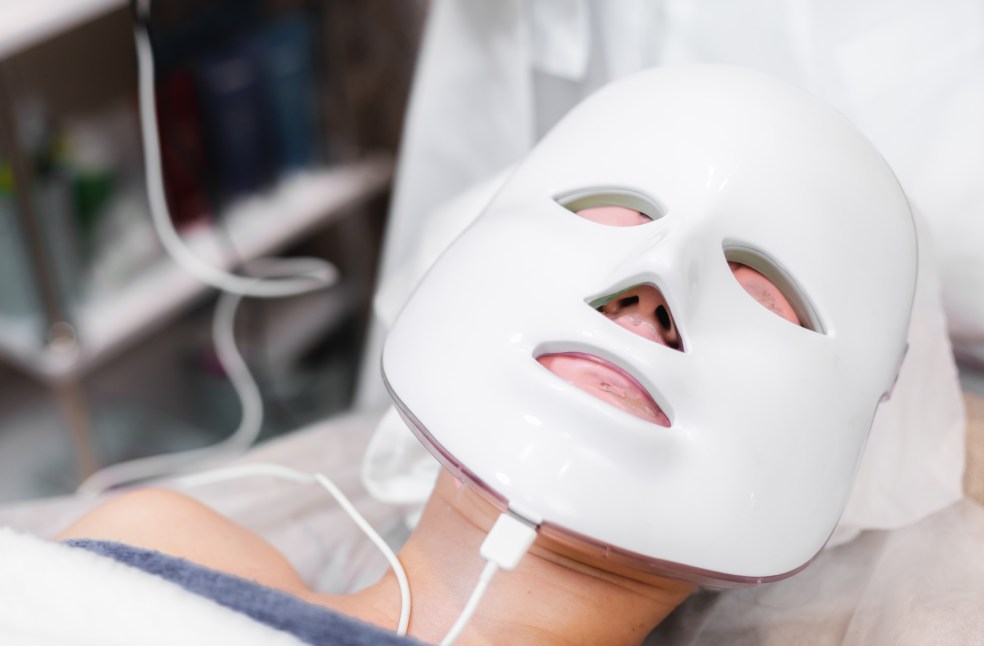
Devices that wish to make medical claims must appear on the MHRA’s Public Access Registration Database (PARD). Dermatologists have also expressed concern, noting that there is a lack of large-scale, long-term clinical studies proving the effectiveness of at-home LED masks.
ASA’s stance
Izzy Dharmasiri from the ASA said adverts can significantly influence purchasing decisions and stressed the need for clear separation between cosmetic benefits and medical claims. Dharmasiri added that evidence must support any advertised claims, especially when vulnerable individuals are seeking genuine solutions to medical issues.
The ASA’s crackdown highlights growing concerns over misleading beauty advertising and reinforces the need for regulator-approved claims particularly when products are promoted as solutions to medical conditions.



