London: A landmark UK study has paved the way for routine screening of children for type 1 diabetes (T1D), with experts saying it could prevent young patients from ‘crash landing’ into a sudden and potentially life-threatening diagnosis.
Following the findings, NHS pre-diabetes clinics for children are set to be introduced after researchers confirmed that simple finger-prick blood tests can detect type 1 diabetes before symptoms appear.
The approach would allow doctors to identify the condition at its earliest stage, well before children become seriously unwell. The study found that children can be diagnosed in the very early stages of type 1 diabetes, a development experts say could lead to a major ‘step change’ in diagnosis and treatment.
Around 400,000 people in the UK are living with type 1 diabetes, representing about 8 percent of all diabetes cases. Currently, around a quarter of children with the condition are diagnosed only after arriving at the hospital in an emergency.

Researchers say early identification could enable children to access treatments that delay the need for insulin for several years, significantly improving outcomes and reducing the shock of an emergency diagnosis.
How the study was carried out
The Early Surveillance for Autoimmune Diabetes (ELSA) study was led by the University of Birmingham and co-funded by Diabetes UK and Breakthrough T1D.
It was designed to assess whether large-scale screening for type 1 diabetes is practical and effective in the UK. Results from the first two years of the study were published as correspondence in The Lancet Diabetes & Endocrinology.
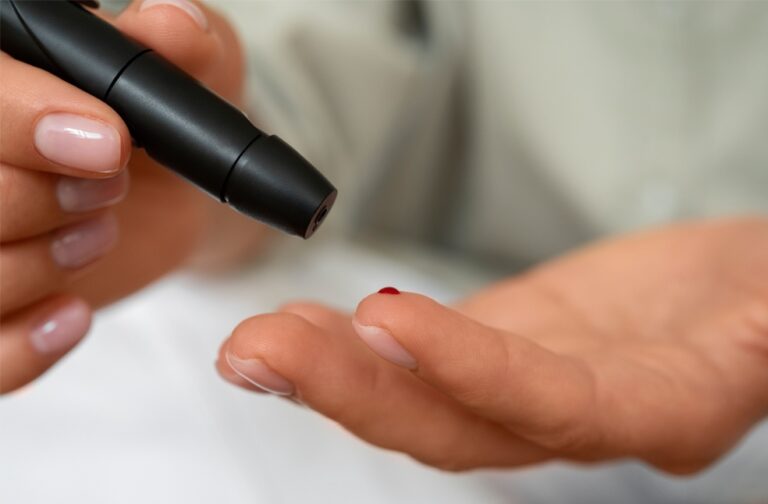
Children aged three to 13 who did not have type 1 diabetes were invited to take part. Participants provided a finger-prick blood sample, which was tested for antibodies previously found in people with pre-symptomatic type 1 diabetes.

Children identified as potentially at risk were invited for further investigations, including additional blood tests or glucose tolerance tests.
In total, 17,283 tests were analysed. More than 200 children were found to be either at risk of developing type 1 diabetes or already showing blood markers indicating an increased risk of the disease.
What happens next
The next phase of the programme, ELSA 2, will expand screening to children aged between two and 17. This phase will support NHS clinics for four years at each of the UK’s 20 study sites.
The clinics will provide education and support to families whose children are found to be at risk of, or in the early stages of, type 1 diabetes. They will also help children as they begin insulin treatment when required.

If approved by the NHS’s spending watchdog, some children may also be offered teplizumab, a treatment approved for use in the UK last year. The drug can delay the need for insulin therapy in people with early-stage type 1 diabetes.
Expert insight
Lead researcher Professor Parth Narendran, Professor of Diabetes Medicine at the University of Birmingham, stated that a future national screening programme could stop children from ‘crash landing’ into a diagnosis.
The Professor explained that treatments such as teplizumab, along with others currently in development, could potentially be offered at this early stage.
“This will mean they don’t need insulin therapy in the long term, and they are kept in the very early stage of type 1 diabetes without insulin requirements. It’s a massive step change,” Prof Narendran said.